之前寫過幾篇降磷藥物的分析,不囉嗦,先整理連結給大家點選複習一下:
1.基礎概念篇,2. 鋁片篇,3. 鈣片篇,4. 自費藥物選擇篇
寶齡生技公司所新研發的拿百磷,2015年1月獲台灣衛福部上市之後,有些需要的腎友已經開始服用,這也是自費降磷藥物的其中一種。
首先,拿百磷在美國的商品名是 Auryxia™(2014年九月美國FDA核准上市),在日本是Riona®,在歐洲是Fexeric®,都和台灣的英文商品名-“Nephoxil”不同,因此,要得到更詳細的研究結果,得用這些名稱去查才行。
拿百磷介紹,正文開始:
主要成分是檸檬酸鐵 (ferric citrate),其中的鐵帶正電(三價),可結合食物中的磷酸(帶負電),不讓腸胃吸收磷,而從大便排出去,達到減少吸收磷的效果,降低洗腎患者的血磷。
由上述可知,這個藥不含鈣,所以身體不會累積鈣,也就減少了血管鈣化的危險。另外,每一公克的拿百磷,含有210毫克的元素鐵,可以增加體內的鐵蛋白(ferritin)含量、運鐵蛋白飽和度(Tsat),有機會改善患者貧血。
可以增加多少?我用美國、日本與台灣的三個研究,來回答這個問題。首先是美國的數據:
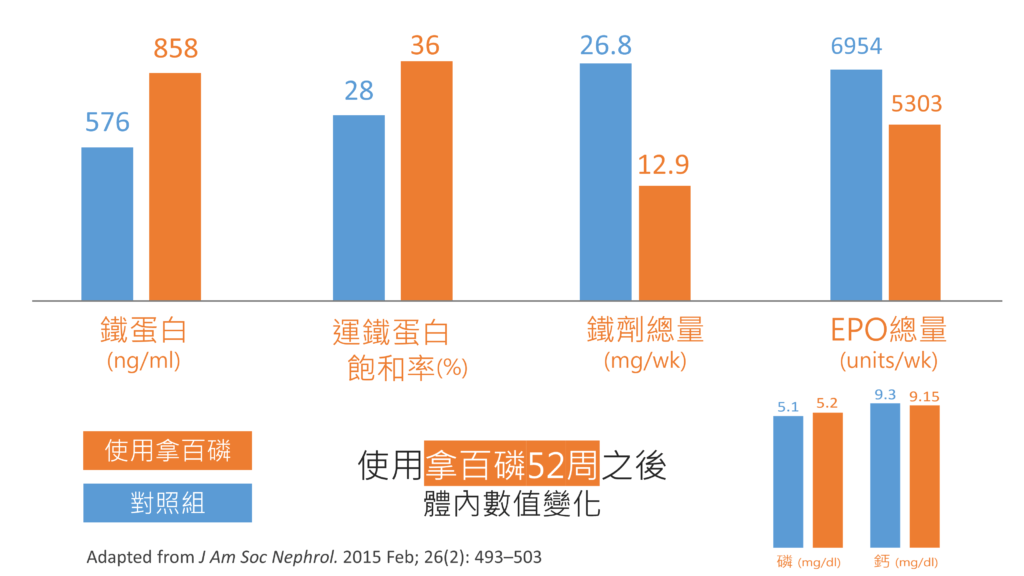
上圖中藍色的對照組,是積極使用鈣片或磷能解的受試者,意思是對照組的患者也積極的使用降磷劑,而結果如圖中所示,使用拿百磷52周(約一年)之後,與鐵相關的數值都上升了,額外補充鐵劑的量減少,紅血球生成素(EPO)的用量也減少了,至於血中鈣與磷(在圖中右下角),算是控制的差不多。
要達到磷控制得宜的這個結果,要吞幾顆呢?
美國的實驗用的是1000毫克的Auryxia®(美國一顆抵兩顆台灣的拿百磷),平均一天要吞8顆,對照組的數量是平均一天7.7顆(鈣片或磷能解)。
接下來是日本研究:
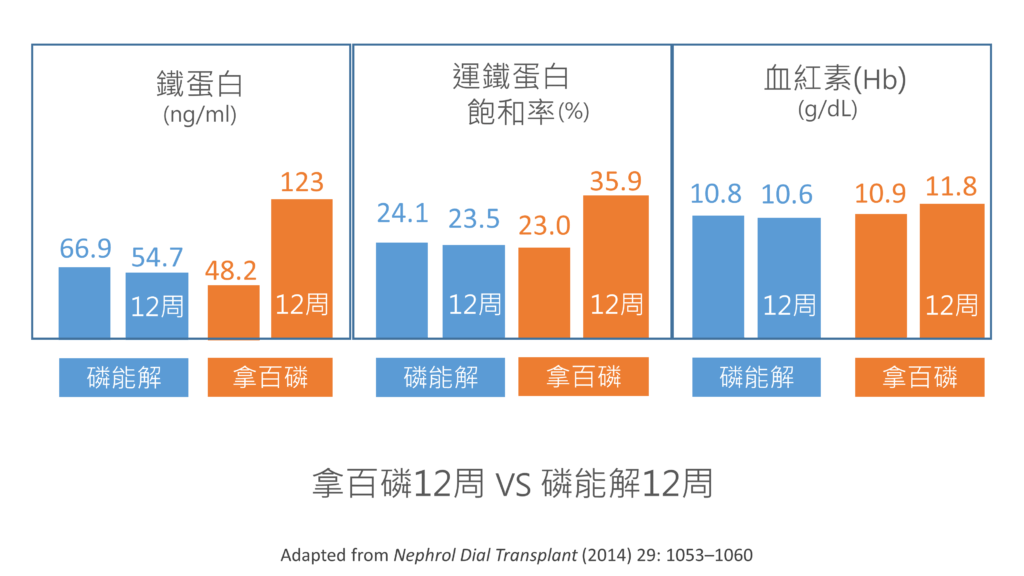
分成兩組患者觀察12個禮拜,一組使用磷能解,另一組使用拿百磷,兩組控制鈣磷的程度差不多,而拿百磷能夠增加一點血紅素。
平均使用拿百磷的劑量為:3.25克/天;而磷能解的劑量平均是:4.94克/天。剛好都是大約六顆。
再來是台灣的研究:
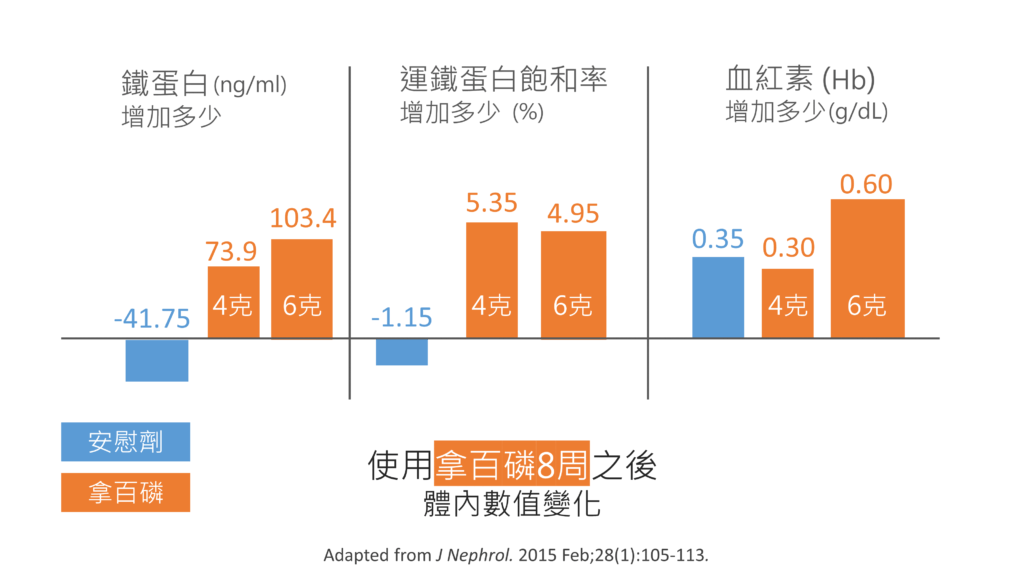
使用8周的拿百磷,除了磷能夠下降1.6 mg/dl (4克/天) 到 2.27 mg/dl (6克/天) 之外,鐵蛋白與血紅素都有增加的趨勢。實驗中所用的劑量:4克,也就是8顆,6克,也就是12顆。
常見的副作用有:大便變黑(發生率約37%)、腹瀉(4-6%)、腹脹或便祕(1-2%)等等。
拿百磷是檸檬酸鐵,鐵是用來結合磷的,那檸檬酸呢?之前我們在講鈣片的時候,有講過不能用檸檬酸鈣,是因為檸檬酸可能會讓食物中的鋁,更容易吸收進體內,造成鋁堆積在體內。2015年的一篇研究指出,經過52周的服用拿百磷,體內血鋁的濃度並未顯著上升(由6.0 µg/L上升到7.0 µg/L),不過呢,體內的鋁並不是只存在於血中,其他部位如:骨頭、肺部、肌肉甚至腦都會有鋁的沉積,因此,這篇研究只抽血驗血中的鋁,不是很能夠說服人,後續還是要小心鋁的後續報導。
總之,這個降磷劑有兩個優點:
一是含鐵,所以體內鐵不足的患者,可以考慮使用。
二是不含鈣,所以體內鈣太高的患者,可以考慮使用。
三個缺點、一個疑慮:
一是含鐵,所以體內鐵太多的,就不能用囉。
二是會有腸胃道症狀、大便變色等等可能的副作用,至於會不會影響腸胃道出血的判斷,我想我可能會蠻頭痛的。
三是如果要吃到有降磷的效果,大概至少一天要吃4克,也就是八顆,也不少顆唷。
疑慮是不曉得會不會增加鋁的吸收,造成體內鋁的堆積,目前沒有強烈證據,就等後續研究結果吧。
聲明:本篇文章純屬學術研究分享,並非廠商贊助所撰寫,藥品使用與否,請與照顧的醫師討論之後,再做決定。
{2147300:JHPWBFXZ};{2147300:JHPWBFXZ},{2147300:BT5V7MHC};{2147300:JHPWBFXZ},{2147300:5UPJNZ26};{2147300:W8FE8TEV},{2147300:T3S3Z4JM},{2147300:DIVD8H7R};{2147300:PARXZ6XN}naturedefaultascno2161%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3A%22zotpress-e38e3302e9e4669fda78cb2c083ffeb1%22%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22W8FE8TEV%22%2C%22library%22%3A%7B%22id%22%3A2147300%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Lee%20et%20al.%22%2C%22parsedDate%22%3A%222015-02%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%202%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ELee%2C%20C.-T.%20%3Ci%3Eet%20al.%3C%5C%2Fi%3E%20Effect%20of%20oral%20ferric%20citrate%20on%20serum%20phosphorus%20in%20hemodialysis%20patients%3A%20multicenter%2C%20randomized%2C%20double-blind%2C%20placebo-controlled%20study.%20%3Ci%3EJ.%20Nephrol.%3C%5C%2Fi%3E%20%3Cb%3E28%3C%5C%2Fb%3E%2C%20105%26%23x2013%3B113%20%282015%29.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Effect%20of%20oral%20ferric%20citrate%20on%20serum%20phosphorus%20in%20hemodialysis%20patients%3A%20multicenter%2C%20randomized%2C%20double-blind%2C%20placebo-controlled%20study%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Chien-Te%22%2C%22lastName%22%3A%22Lee%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22I.-Wen%22%2C%22lastName%22%3A%22Wu%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Shou-Shan%22%2C%22lastName%22%3A%22Chiang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yu-Sen%22%2C%22lastName%22%3A%22Peng%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kuo-Hsiung%22%2C%22lastName%22%3A%22Shu%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ming-Ju%22%2C%22lastName%22%3A%22Wu%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mai-Szu%22%2C%22lastName%22%3A%22Wu%22%7D%5D%2C%22abstractNote%22%3A%22BACKGROUND%3A%20Hyperphosphatemia%20is%20a%20common%20complication%20in%20dialysis%20patients%20that%20can%20be%20treated%20by%20oral%20phosphate%20binders.%20We%20investigated%20the%20efficacy%20and%20safety%20of%20oral%20ferric%20citrate%20as%20a%20phosphate%20binder%20for%20Taiwanese%20patients%20with%20end%20stage%20renal%20disease%20and%20with%20hyperphosphatemia%20who%20were%20undergoing%20hemodialysis.%5CnMETHODS%3A%20This%20was%20a%20prospective%2C%20double-blind%2C%20placebo-controlled%2C%20randomized%20trial%20carried%20out%20in%205%20hospitals%20in%20Taiwan.%20Ferric%20citrate%20%284%20or%206%20g%5C%2Fday%29%20or%20placebo%20was%20administered%20for%2056%20days.%20Serum%20calcium%2C%20phosphorous%20levels%2C%20calcium%20%5Cu00d7%20phosphorus%20product%2C%20serum%20ferritin%20level%2C%20transferrin%20saturation%2C%20and%20adverse%20events%20were%20recorded.%5CnRESULTS%3A%20A%20total%20of%20166%20patients%20completed%20the%20trial.%20The%20placebo%20group%20had%20relatively%20constant%20serum%20data.%20Serum%20phosphorus%20declined%20significantly%20in%20the%206%20g%5C%2Fday%20group%20%28p%20%3C%200.05%20for%204%20and%208%20weeks%29%20and%20the%204%20g%5C%2Fday%20group%20%28p%20%3C%200.05%20for%204%20and%208%20weeks%29.%20There%20were%20similar%20changes%20in%20the%20calcium%20%5Cu00d7%20phosphorus%20product.%20The%20serum%20ferritin%20level%20increased%20significantly%20in%20the%206%20g%5C%2Fday%20group%20%28p%20%3C%200.05%29%20and%20the%204%20g%5C%2Fday%20group%20%28p%20%3C%200.05%29.%20There%20were%20similar%20changes%20in%20transferrin%20saturation.%20Most%20adverse%20events%20were%20mild%20to%20moderate%20and%20were%20comparable%20among%20the%20three%20groups.%5CnCONCLUSIONS%3A%20A%2056-day%20treatment%20with%20ferric%20citrate%20effectively%20controlled%20hyperphosphatemia%20and%20was%20well%20tolerated%20in%20maintenance%20hemodialysis%20patients.%20There%20were%20also%20moderate%20increases%20in%20serum%20ferritin%20and%20transferrin%20saturation.%22%2C%22date%22%3A%22Feb%202015%22%2C%22language%22%3A%22ENG%22%2C%22DOI%22%3A%2210.1007%5C%2Fs40620-014-0108-6%22%2C%22ISSN%22%3A%221724-6059%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222016-12-02T02%3A12%3A03Z%22%7D%7D%2C%7B%22key%22%3A%22DIVD8H7R%22%2C%22library%22%3A%7B%22id%22%3A2147300%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Yokoyama%20et%20al.%22%2C%22parsedDate%22%3A%222014-05%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%202%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EYokoyama%2C%20K.%20%3Ci%3Eet%20al.%3C%5C%2Fi%3E%20A%20randomized%20trial%20of%20JTT-751%20versus%20sevelamer%20hydrochloride%20in%20patients%20on%20hemodialysis.%20%3Ci%3ENephrol.%20Dial.%20Transplant.%3C%5C%2Fi%3E%20%3Cb%3E29%3C%5C%2Fb%3E%2C%201053%26%23x2013%3B1060%20%282014%29.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22A%20randomized%20trial%20of%20JTT-751%20versus%20sevelamer%20hydrochloride%20in%20patients%20on%20hemodialysis%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Keitaro%22%2C%22lastName%22%3A%22Yokoyama%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Takashi%22%2C%22lastName%22%3A%22Akiba%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masafumi%22%2C%22lastName%22%3A%22Fukagawa%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masaaki%22%2C%22lastName%22%3A%22Nakayama%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenichi%22%2C%22lastName%22%3A%22Sawada%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yuji%22%2C%22lastName%22%3A%22Kumagai%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Glenn%20M.%22%2C%22lastName%22%3A%22Chertow%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hideki%22%2C%22lastName%22%3A%22Hirakata%22%7D%5D%2C%22abstractNote%22%3A%22BACKGROUND%3A%20JTT-751%20is%20a%20novel%20phosphate%20binder%20containing%20ferric%20citrate%20as%20the%20active%20ingredient.%5CnMETHODS%3A%20In%20this%20Phase%203%2C%20multicenter%2C%20randomized%2C%20open-label%2C%20parallel-group%20study%2C%20we%20compared%20the%20efficacy%20and%20safety%20of%20JTT-751%20and%20sevelamer%20hydrochloride%20in%20patients%20undergoing%20hemodialysis.%20A%20total%20of%20230%20patients%20with%20a%20serum%20phosphate%20%5Cu22651.97%20and%20%3C3.23%20mmol%5C%2FL%20were%20randomized%20to%20JTT-751%20%28dose%20adjusted%20between%201.5%20and%206.0%20g%5C%2Fday%29%20or%20sevelamer%20hydrochloride%20%28dose%20adjusted%20between%203.0%20and%209.0%20g%5C%2Fday%29%20for%2012%20weeks.%20The%20primary%20outcome%20was%20change%20in%20serum%20phosphate%20from%20baseline%20to%20end%20of%20treatment.%20Secondary%20outcomes%20included%20the%20changes%20in%20corrected%20serum%20calcium%20and%20intact%20parathyroid%20hormone%20%28PTH%29.%20The%20changes%20in%20ferritin%2C%20transferrin%20saturation%20and%20erythropoiesis-stimulating%20agent%20dose%20were%20additional%20outcomes.%5CnRESULTS%3A%20Changes%20in%20serum%20phosphate%20at%20the%20end%20of%20treatment%20were%20-0.82%20mmol%5C%2FL%20in%20the%20JTT-751%20group%20and%20-0.78%20mmol%5C%2FL%20in%20the%20sevelamer%20group%2C%20establishing%20non-inferiority%20of%20JTT-751%20compared%20with%20sevelamer%20%28least%20squares%20mean%2C%20-0.03%20mmol%5C%2FL%3B%2095%25%20confidence%20interval%2C%20-0.13%20to%200.07%20mmol%5C%2FL%29.%20Corrected%20serum%20calcium%20increased%20and%20PTH%20decreased%20from%20baseline%20within%20both%20groups%3B%20changes%20between%20groups%20were%20similar.%20Gastrointestinal%20disorders%20were%20the%20most%20common%20adverse%20events%20in%20both%20groups%3B%20the%20incidence%20of%20diarrhea%20was%20higher%20in%20the%20JTT-751%20group%2C%20while%20constipation%20occurred%20frequently%20in%20the%20sevelamer%20group.%20Treatment%20with%20JTT-751%20resulted%20in%20significant%20relative%20increases%20in%20serum%20ferritin%20and%20transferrin%20saturation.%5CnCONCLUSIONS%3A%20Efficacy%20and%20safety%20of%20JTT-751%20was%20comparable%20to%20sevelamer%20in%20patients%20on%20hemodialysis%20with%20hyperphosphatemia.%20Differential%20adverse%20effects%20were%20observed%3B%20biochemical%20markers%20of%20iron%20status%20increased%20in%20patients%20treated%20with%20JTT-751.%5CnTRIAL%20REGISTRATION%20NUMBER%3A%20CTI-111433%20%28The%20Japan%20Pharmaceutical%20Information%20Center%20at%3A%20http%5C%2F%5C%2Fwww.clinicaltrials.jp%29.%20Date%20of%20registration%3A%207%20March%202011.%22%2C%22date%22%3A%22May%202014%22%2C%22language%22%3A%22ENG%22%2C%22DOI%22%3A%2210.1093%5C%2Fndt%5C%2Fgft483%22%2C%22ISSN%22%3A%221460-2385%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222016-12-02T01%3A55%3A59Z%22%7D%7D%2C%7B%22key%22%3A%22T3S3Z4JM%22%2C%22library%22%3A%7B%22id%22%3A2147300%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Lewis%20et%20al.%22%2C%22parsedDate%22%3A%222015-02%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%202%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ELewis%2C%20J.%20B.%20%3Ci%3Eet%20al.%3C%5C%2Fi%3E%20Ferric%20Citrate%20Controls%20Phosphorus%20and%20Delivers%20Iron%20in%20Patients%20on%20Dialysis.%20%3Ci%3EJ%20Am%20Soc%20Nephrol%3C%5C%2Fi%3E%20%3Cb%3E26%3C%5C%2Fb%3E%2C%20493%26%23x2013%3B503%20%282015%29.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Ferric%20Citrate%20Controls%20Phosphorus%20and%20Delivers%20Iron%20in%20Patients%20on%20Dialysis%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Julia%20B.%22%2C%22lastName%22%3A%22Lewis%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mohammed%22%2C%22lastName%22%3A%22Sika%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mark%20J.%22%2C%22lastName%22%3A%22Koury%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Peale%22%2C%22lastName%22%3A%22Chuang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gerald%22%2C%22lastName%22%3A%22Schulman%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mark%20T.%22%2C%22lastName%22%3A%22Smith%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Frederick%20C.%22%2C%22lastName%22%3A%22Whittier%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Douglas%20R.%22%2C%22lastName%22%3A%22Linfert%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Claude%20M.%22%2C%22lastName%22%3A%22Galphin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Balaji%20P.%22%2C%22lastName%22%3A%22Athreya%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%20Kaldun%20Kaldun%22%2C%22lastName%22%3A%22Nossuli%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ingrid%20J.%22%2C%22lastName%22%3A%22Chang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Samuel%20S.%22%2C%22lastName%22%3A%22Blumenthal%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22John%22%2C%22lastName%22%3A%22Manley%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Steven%22%2C%22lastName%22%3A%22Zeig%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kotagal%20S.%22%2C%22lastName%22%3A%22Kant%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Juan%20Jose%22%2C%22lastName%22%3A%22Olivero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tom%22%2C%22lastName%22%3A%22Greene%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jamie%20P.%22%2C%22lastName%22%3A%22Dwyer%22%7D%5D%2C%22abstractNote%22%3A%22Patients%20on%20dialysis%20require%20phosphorus%20binders%20to%20prevent%20hyperphosphatemia%20and%20are%20iron%20deficient.%20We%20studied%20ferric%20citrate%20as%20a%20phosphorus%20binder%20and%20iron%20source.%20In%20this%20sequential%2C%20randomized%20trial%2C%20441%20subjects%20on%20dialysis%20were%20randomized%20to%20ferric%20citrate%20or%20active%20control%20in%20a%2052-week%20active%20control%20period%20followed%20by%20a%204-week%20placebo%20control%20period%2C%20in%20which%20subjects%20on%20ferric%20citrate%20who%20completed%20the%20active%20control%20period%20were%20rerandomized%20to%20ferric%20citrate%20or%20placebo.%20The%20primary%20analysis%20compared%20the%20mean%20change%20in%20phosphorus%20between%20ferric%20citrate%20and%20placebo%20during%20the%20placebo%20control%20period.%20A%20sequential%20gatekeeping%20strategy%20controlled%20study-wise%20type%201%20error%20for%20serum%20ferritin%2C%20transferrin%20saturation%2C%20and%20intravenous%20iron%20and%20erythropoietin-stimulating%20agent%20usage%20as%20prespecified%20secondary%20outcomes%20in%20the%20active%20control%20period.%20Ferric%20citrate%20controlled%20phosphorus%20compared%20with%20placebo%2C%20with%20a%20mean%20treatment%20difference%20of%20%5Cu22122.2%5Cu00b10.2%20mg%5C%2Fdl%20%28mean%5Cu00b1SEM%29%20%28P%3C0.001%29.%20Active%20control%20period%20phosphorus%20was%20similar%20between%20ferric%20citrate%20and%20active%20control%2C%20with%20comparable%20safety%20profiles.%20Subjects%20on%20ferric%20citrate%20achieved%20higher%20mean%20iron%20parameters%20%28ferritin%3D899%5Cu00b1488%20ng%5C%2Fml%20%5Bmean%5Cu00b1SD%5D%3B%20transferrin%20saturation%3D39%25%5Cu00b117%25%29%20versus%20subjects%20on%20active%20control%20%28ferritin%3D628%5Cu00b1367%20ng%5C%2Fml%20%5Bmean%5Cu00b1SD%5D%3B%20transferrin%20saturation%3D30%25%5Cu00b112%25%3B%20P%3C0.001%20for%20both%29.%20Subjects%20on%20ferric%20citrate%20received%20less%20intravenous%20elemental%20iron%20%28median%3D12.95%20mg%5C%2Fwk%20ferric%20citrate%3B%2026.88%20mg%5C%2Fwk%20active%20control%3B%20P%3C0.001%29%20and%20less%20erythropoietin-stimulating%20agent%20%28median%20epoetin-equivalent%20units%20per%20week%3A%205306%20units%5C%2Fwk%20ferric%20citrate%3B%206951%20units%5C%2Fwk%20active%20control%3B%20P%3D0.04%29.%20Hemoglobin%20levels%20were%20statistically%20higher%20on%20ferric%20citrate.%20Thus%2C%20ferric%20citrate%20is%20an%20efficacious%20and%20safe%20phosphate%20binder%20that%20increases%20iron%20stores%20and%20reduces%20intravenous%20iron%20and%20erythropoietin-stimulating%20agent%20use%20while%20maintaining%20hemoglobin.%22%2C%22date%22%3A%222015-2%22%2C%22language%22%3A%22%22%2C%22DOI%22%3A%2210.1681%5C%2FASN.2014020212%22%2C%22ISSN%22%3A%221046-6673%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fwww.ncbi.nlm.nih.gov%5C%2Fpmc%5C%2Farticles%5C%2FPMC4310662%5C%2F%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222016-12-02T00%3A51%3A54Z%22%7D%7D%2C%7B%22key%22%3A%225UPJNZ26%22%2C%22library%22%3A%7B%22id%22%3A2147300%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Pennoyer%20and%20Bridgeman%22%2C%22parsedDate%22%3A%222015-05%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%202%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EPennoyer%2C%20A.%20%26amp%3B%20Bridgeman%2C%20M.%20B.%20Ferric%20Citrate%20%28Auryxia%29%20for%20the%20Treatment%20of%20Hyperphosphatemia.%20%3Ci%3EP%20T%3C%5C%2Fi%3E%20%3Cb%3E40%3C%5C%2Fb%3E%2C%20329%26%23x2013%3B339%20%282015%29.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Ferric%20Citrate%20%28Auryxia%29%20for%20the%20Treatment%20of%20Hyperphosphatemia%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Adam%22%2C%22lastName%22%3A%22Pennoyer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mary%20Barna%22%2C%22lastName%22%3A%22Bridgeman%22%7D%5D%2C%22abstractNote%22%3A%22Ferric%20citrate%20%28Auryxia%29%20for%20the%20treatment%20of%20hyperphosphatemia%22%2C%22date%22%3A%222015-5%22%2C%22language%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22ISSN%22%3A%221052-1372%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fwww.ncbi.nlm.nih.gov%5C%2Fpmc%5C%2Farticles%5C%2FPMC4422633%5C%2F%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222016-11-29T02%3A22%3A05Z%22%7D%7D%2C%7B%22key%22%3A%22JHPWBFXZ%22%2C%22library%22%3A%7B%22id%22%3A2147300%7D%2C%22meta%22%3A%7B%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%202%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EPBF%26%23x5BF6%3B%26%23x9F61%3B%26%23x5BCC%3B%26%23x9326%3B%26%23x751F%3B%26%23x6280%3B.%20%3Ca%20href%3D%27http%3A%5C%2F%5C%2Fwww.pbf.com.tw%5C%2Fcht%5C%2Fresearch.php%3Fserial%3D1%27%3Ehttp%3A%5C%2F%5C%2Fwww.pbf.com.tw%5C%2Fcht%5C%2Fresearch.php%3Fserial%3D1%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22PBF%5Cu5bf6%5Cu9f61%5Cu5bcc%5Cu9326%5Cu751f%5Cu6280%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fwww.pbf.com.tw%5C%2Fcht%5C%2Fresearch.php%3Fserial%3D1%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%228V4TNTHP%22%5D%2C%22dateModified%22%3A%222016-11-29T02%3A15%3A25Z%22%7D%7D%2C%7B%22key%22%3A%22BT5V7MHC%22%2C%22library%22%3A%7B%22id%22%3A2147300%7D%2C%22meta%22%3A%7B%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%202%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EAuryxia%2C%20Fexeric%2C%20Riona%2C%20ferric%20citrate%20%28JTT-751%2C%20KRX-0502%29%20-%20Product%20Profile%20-%20BioCentury.%20%3Ca%20href%3D%27http%3A%5C%2F%5C%2Fwww.biocentury.com%5C%2Fproducts%5C%2Fauryxia%27%3Ehttp%3A%5C%2F%5C%2Fwww.biocentury.com%5C%2Fproducts%5C%2Fauryxia%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Auryxia%2C%20Fexeric%2C%20Riona%2C%20ferric%20citrate%20%28JTT-751%2C%20KRX-0502%29%20-%20Product%20Profile%20-%20BioCentury%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fwww.biocentury.com%5C%2Fproducts%5C%2Fauryxia%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222016-11-29T02%3A11%3A58Z%22%7D%7D%2C%7B%22key%22%3A%22PARXZ6XN%22%2C%22library%22%3A%7B%22id%22%3A2147300%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Van%20Buren%20et%20al.%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%202%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EVan%20Buren%2C%20P.%20N.%20%3Ci%3Eet%20al.%3C%5C%2Fi%3E%20The%20Phosphate%20Binder%20Ferric%20Citrate%20and%20Mineral%20Metabolism%20and%20Inflammatory%20Markers%20in%20Maintenance%20Dialysis%20Patients%3A%20Results%20From%20Prespecified%20Analyses%20of%20a%20Randomized%20Clinical%20Trial.%20%3Ci%3EAmerican%20Journal%20of%20Kidney%20Diseases%3C%5C%2Fi%3E%20%3Ca%20href%3D%27http%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1053%5C%2Fj.ajkd.2015.03.013%27%3Ehttp%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1053%5C%2Fj.ajkd.2015.03.013%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22The%20Phosphate%20Binder%20Ferric%20Citrate%20and%20Mineral%20Metabolism%20and%20Inflammatory%20Markers%20in%20Maintenance%20Dialysis%20Patients%3A%20Results%20From%20Prespecified%20Analyses%20of%20a%20Randomized%20Clinical%20Trial%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Peter%20N.%22%2C%22lastName%22%3A%22Van%20Buren%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Julia%20B.%22%2C%22lastName%22%3A%22Lewis%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jamie%20P.%22%2C%22lastName%22%3A%22Dwyer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tom%22%2C%22lastName%22%3A%22Greene%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22John%22%2C%22lastName%22%3A%22Middleton%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mohammed%22%2C%22lastName%22%3A%22Sika%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kausik%22%2C%22lastName%22%3A%22Umanath%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Josephine%20D.%22%2C%22lastName%22%3A%22Abraham%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Shahabul%20S.%22%2C%22lastName%22%3A%22Arfeen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Isai%20G.%22%2C%22lastName%22%3A%22Bowline%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gil%22%2C%22lastName%22%3A%22Chernin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Stephen%20Z.%22%2C%22lastName%22%3A%22Fadem%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Simin%22%2C%22lastName%22%3A%22Goral%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mark%22%2C%22lastName%22%3A%22Koury%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Marvin%20V.%22%2C%22lastName%22%3A%22Sinsakul%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Daniel%20E.%22%2C%22lastName%22%3A%22Weiner%22%7D%5D%2C%22abstractNote%22%3A%22Background%5CnPhosphate%20binders%20are%20the%20cornerstone%20of%20hyperphosphatemia%20management%20in%20dialysis%20patients.%20Ferric%20citrate%20is%20an%20iron-based%20oral%20phosphate%20binder%20that%20effectively%20lowers%20serum%20phosphorus%20levels.%5CnStudy%20Design%5Cn52-week%2C%20open-label%2C%20phase%203%2C%20randomized%2C%20controlled%20trial%20for%20safety-profile%20assessment.%5CnSetting%20%26amp%3B%20Participants%5CnMaintenance%20dialysis%20patients%20with%20serum%20phosphorus%20levels%5Cu00a0%5Cu22656.0%20mg%5C%2FdL%20after%20washout%20of%20prior%20phosphate%20binders.%5CnIntervention%5Cn2%3A1%20randomization%20to%20ferric%20citrate%20or%20active%20control%20%28sevelamer%20carbonate%20and%5C%2For%20calcium%20acetate%29.%5CnOutcomes%5CnChanges%20in%20mineral%20bone%20disease%2C%20protein-energy%20wasting%5C%2Finflammation%2C%20and%20occurrence%20of%20adverse%20events%20after%201%20year.%5CnMeasurements%5CnSerum%20calcium%2C%20intact%20parathyroid%20hormone%2C%20phosphorus%2C%20aluminum%2C%20white%20blood%20cell%20count%2C%20percentage%20of%20lymphocytes%2C%20serum%20urea%20nitrogen%2C%20and%20bicarbonate.%5CnResults%5CnThere%20were%20292%20participants%20randomly%20assigned%20to%20ferric%20citrate%2C%20and%20149%2C%20to%20active%20control.%20Groups%20were%20well%20matched.%20For%20mean%20changes%20from%20baseline%2C%20phosphorus%20levels%20decreased%20similarly%20in%20the%20ferric%20citrate%20and%20active%20control%20groups%20%28%5Cu22122.04%20%5Cu00b1%201.99%20%5BSD%5D%20vs%5Cu00a0%5Cu22122.18%20%5Cu00b1%202.25%20mg%5C%2FdL%2C%20respectively%3B%20P%20%3D%200.9%29%3B%20serum%20calcium%20levels%20increased%20similarly%20in%20the%20ferric%20citrate%20and%20active%20control%20groups%20%280.22%20%5Cu00b1%200.90%20vs%200.31%20%5Cu00b1%200.95%20mg%5C%2FdL%3B%20P%20%3D%200.2%29.%20Hypercalcemia%20occurred%20in%204%20participants%20receiving%20calcium%20acetate.%20Parathyroid%20hormone%20levels%20decreased%20similarly%20in%20the%20ferric%20citrate%20and%20active%20control%20groups%20%28%5Cu2212167.1%20%5Cu00b1%20399.8%20vs%5Cu00a0%5Cu2212152.7%20%5Cu00b1%20392.1%20pg%5C%2FmL%3B%20P%20%3D%200.8%29.%20Serum%20albumin%2C%20bicarbonate%2C%20serum%20urea%20nitrogen%2C%20white%20blood%20cell%20count%20and%20percentage%20of%20lymphocytes%2C%20and%20aluminum%20values%20were%20similar%20between%20ferric%20citrate%20and%20active%20control.%20Total%20and%20low-density%20lipoprotein%20cholesterol%20levels%20were%20lower%20in%20participants%20receiving%20sevelamer%20than%20those%20receiving%20ferric%20citrate%20and%20calcium%20acetate.%20Fewer%20participants%20randomly%20assigned%20to%20ferric%20citrate%20had%20serious%20adverse%20events%20compared%20with%20active%20control.%5CnLimitations%5CnOpen-label%20study%2C%20few%20peritoneal%20dialysis%20patients.%5CnConclusions%5CnFerric%20citrate%20was%20associated%20with%20similar%20phosphorus%20control%20compared%20to%20active%20control%2C%20with%20similar%20effects%20on%20markers%20of%20bone%20and%20mineral%20metabolism%20in%20dialysis%20patients.%20There%20was%20no%20evidence%20of%20protein-energy%20wasting%5C%2Finflammation%20or%20aluminum%20toxicity%2C%20and%20fewer%20participants%20randomly%20assigned%20to%20ferric%20citrate%20had%20serious%20adverse%20events.%20Ferric%20citrate%20is%20an%20effective%20phosphate%20binder%20with%20a%20safety%20profile%20comparable%20to%20sevelamer%20and%20calcium%20acetate.%22%2C%22date%22%3A%22%22%2C%22language%22%3A%22%22%2C%22DOI%22%3A%2210.1053%5C%2Fj.ajkd.2015.03.013%22%2C%22ISSN%22%3A%220272-6386%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fwww.sciencedirect.com%5C%2Fscience%5C%2Farticle%5C%2Fpii%5C%2FS0272638615005417%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222015-06-03T08%3A06%3A09Z%22%7D%7D%5D%7D 1.
Lee, C.-T. et al. Effect of oral ferric citrate on serum phosphorus in hemodialysis patients: multicenter, randomized, double-blind, placebo-controlled study. J. Nephrol. 28, 105–113 (2015).
1.
Yokoyama, K. et al. A randomized trial of JTT-751 versus sevelamer hydrochloride in patients on hemodialysis. Nephrol. Dial. Transplant. 29, 1053–1060 (2014).
1.
Lewis, J. B. et al. Ferric Citrate Controls Phosphorus and Delivers Iron in Patients on Dialysis. J Am Soc Nephrol 26, 493–503 (2015).
1.
Pennoyer, A. & Bridgeman, M. B. Ferric Citrate (Auryxia) for the Treatment of Hyperphosphatemia. P T 40, 329–339 (2015).
1.
Van Buren, P. N.
et al. The Phosphate Binder Ferric Citrate and Mineral Metabolism and Inflammatory Markers in Maintenance Dialysis Patients: Results From Prespecified Analyses of a Randomized Clinical Trial.
American Journal of Kidney Diseases http://doi.org/10.1053/j.ajkd.2015.03.013.