美國FDA 通過了第一個「能夠治療“類澱粉沉積症”的吸附器材」Lixelle™
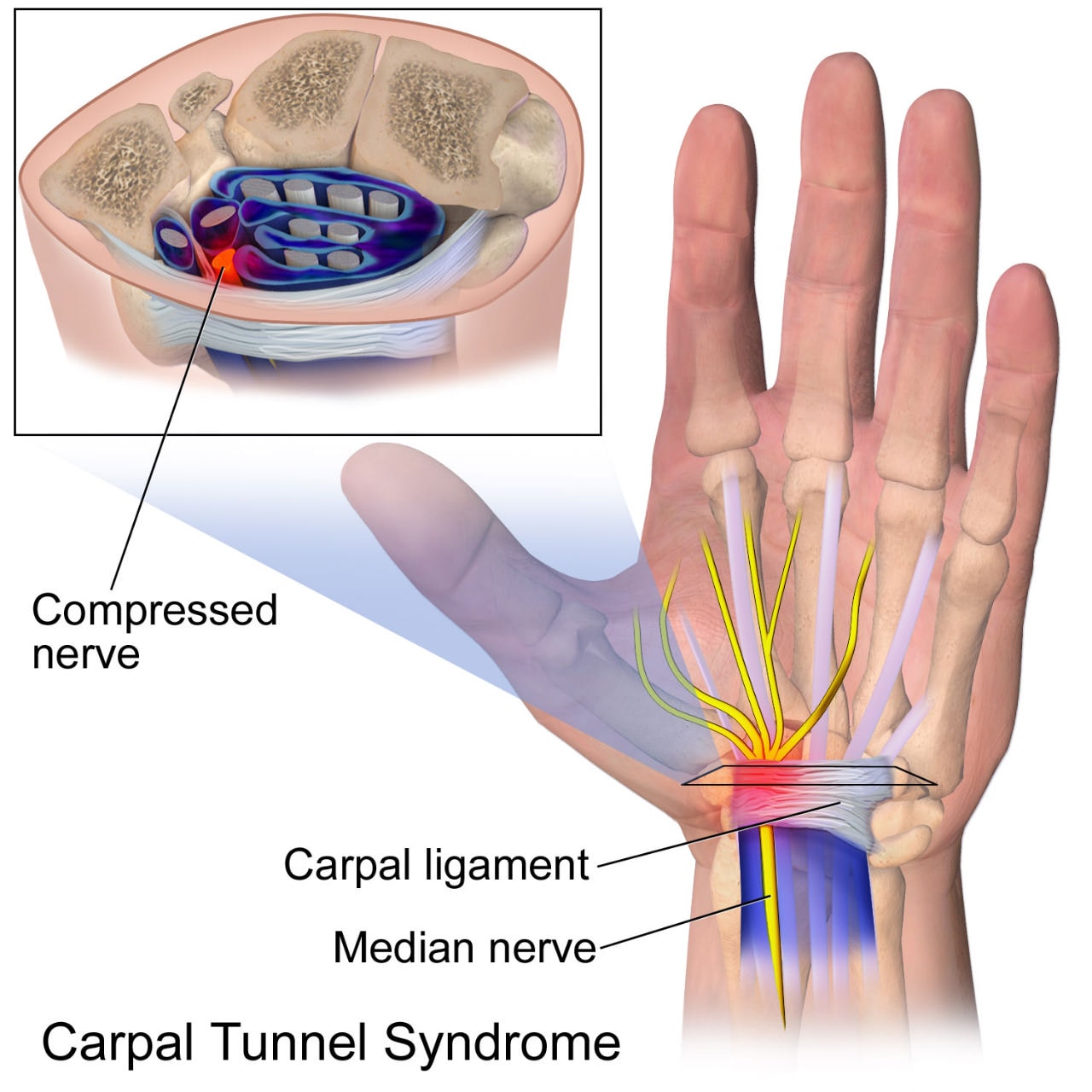
“類澱粉沉積症”常常困擾著長期洗腎患者,因為血中的某些分子無法透析乾淨,導至日積月累的陳積在體內,尤其是洗超過五年、超過60歲的患者。常見的症狀有:手指(大拇指到無名指)的麻木–腕隧道症候群,關節疼痛–類澱粉沉積所造成的關節發炎,骨囊腫(bone cyst),destructive spondyloarthropathy等等。
這個日本人做的器材,是以串聯的方式,接在人工腎臟之前,讓透析患者的血液,先通過這個器材(Lixelle™)吸附Beta-2 microglobulin,再經過人工腎臟去做常規的透析。
用PDA文中的描述去搜尋,跳出來的產品網址是:(台灣目前沒有啦) http://www.kaneka-med.jp/english/products/apheresis/px_001c/
文中還有提到,常見副作用有:透析中低血壓,以及血比容下降。但是這也是血液透析患者常見的問題,所以需要產品公司注意後續副作用的比例,來確保進一步的安全性。
去搜尋了一下關於Lixelle™的研究,果然還是日本的研究最多,有一個研究是:17個確認有“類澱粉沉積症”的洗腎患者,先用一般high-flux(高通透性)的人工腎臟透析一年,之後再用Lixelle™加上high-flux的人工腎臟一起使用一年,結果顯示:一般high-flux人工腎臟使用了一年後,體內的Beta-2 microglobulin濃度沒有改變,而加上Lixelle™之後,不僅血中Beta-2 microglobulin濃度顯著下降,手指抓握力氣、神經傳導速度(median motor terminal latency)、以及日常生活情況都有顯著改善。
結論:看起來是個好東西啊,但引進台灣後,負擔的起的人不知道有多少呢…
{2147300:75IASB8J};{2147300:Z8H4VA8C};{2147300:75IASB8J};{2147300:FUHCAWJM}pediatricsdefaultascno584%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3A%22zotpress-41649ab662a90e8f74c5a721a9e759a4%22%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22FUHCAWJM%22%2C%22library%22%3A%7B%22id%22%3A2147300%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Abe%20et%20al.%22%2C%22parsedDate%22%3A%222003-10%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EAbe%20T%2C%20Uchita%20K%2C%20Orita%20H%2C%20et%20al.%20Effect%20of%20%26%23x3B2%3B2-microglobulin%20adsorption%20column%20on%20dialysis-related%20amyloidosis.%20%3Ci%3EKidney%20Int%3C%5C%2Fi%3E.%202003%3B64%284%29%3A1522-1528.%20%3Ca%20href%3D%27http%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1046%5C%2Fj.1523-1755.2003.00235.x%27%3Ehttp%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1046%5C%2Fj.1523-1755.2003.00235.x%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Effect%20of%20%5Cu03b22-microglobulin%20adsorption%20column%20on%20dialysis-related%20amyloidosis%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Takaya%22%2C%22lastName%22%3A%22Abe%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kazuhiro%22%2C%22lastName%22%3A%22Uchita%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hikari%22%2C%22lastName%22%3A%22Orita%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Motohiro%22%2C%22lastName%22%3A%22Kamimura%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Minoru%22%2C%22lastName%22%3A%22Oda%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hirohito%22%2C%22lastName%22%3A%22Hasegawa%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hirotsugu%22%2C%22lastName%22%3A%22Kobata%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masaaki%22%2C%22lastName%22%3A%22Fukunishi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masami%22%2C%22lastName%22%3A%22Shimazaki%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tomiya%22%2C%22lastName%22%3A%22Abe%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tadao%22%2C%22lastName%22%3A%22Akizawa%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Suhail%22%2C%22lastName%22%3A%22Ahmad%22%7D%5D%2C%22abstractNote%22%3A%22Effect%20of%202-microglobulin%20adsorption%20column%20on%20dialysis-related%20amyloidosis.%22%2C%22date%22%3A%22October%202003%22%2C%22language%22%3A%22en%22%2C%22DOI%22%3A%2210.1046%5C%2Fj.1523-1755.2003.00235.x%22%2C%22ISSN%22%3A%220085-2538%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fwww.nature.com%5C%2Fki%5C%2Fjournal%5C%2Fv64%5C%2Fn4%5C%2Ffull%5C%2F4494048a.html%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222015-03-21T07%3A33%3A46Z%22%7D%7D%2C%7B%22key%22%3A%22Z8H4VA8C%22%2C%22library%22%3A%7B%22id%22%3A2147300%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Yamamoto%20et%20al.%22%2C%22parsedDate%22%3A%222013-06-12%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EYamamoto%20S%2C%20James%20J%2C%20Maruyama%20H%2C%20Narit%20I.%20Dialysis-Related%20Amyloidosis%3A%20Pathogenesis%20and%20Clinical%20Features%20in%20Patients%20Undergoing%20Dialysis%20Treatment.%20In%3A%20Feng%20D%2C%20ed.%20%3Ci%3EAmyloidosis%3C%5C%2Fi%3E.%20InTech%3B%202013.%20Accessed%20March%2021%2C%202015.%20%3Ca%20href%3D%27http%3A%5C%2F%5C%2Fwww.intechopen.com%5C%2Fbooks%5C%2Famyloidosis%5C%2Fdialysis-related-amyloidosis-pathogenesis-and-clinical-features-in-patients-undergoing-dialysis-trea%27%3Ehttp%3A%5C%2F%5C%2Fwww.intechopen.com%5C%2Fbooks%5C%2Famyloidosis%5C%2Fdialysis-related-amyloidosis-pathogenesis-and-clinical-features-in-patients-undergoing-dialysis-trea%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22bookSection%22%2C%22title%22%3A%22Dialysis-Related%20Amyloidosis%3A%20Pathogenesis%20and%20Clinical%20Features%20in%20Patients%20Undergoing%20Dialysis%20Treatment%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22editor%22%2C%22firstName%22%3A%22Dali%22%2C%22lastName%22%3A%22Feng%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Suguru%22%2C%22lastName%22%3A%22Yamamoto%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Junichiro%22%2C%22lastName%22%3A%22James%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hiroki%22%2C%22lastName%22%3A%22Maruyama%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ichiei%22%2C%22lastName%22%3A%22Narit%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22bookTitle%22%3A%22Amyloidosis%22%2C%22date%22%3A%222013-06-12%22%2C%22language%22%3A%22en%22%2C%22ISBN%22%3A%22978-953-51-1100-9%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fwww.intechopen.com%5C%2Fbooks%5C%2Famyloidosis%5C%2Fdialysis-related-amyloidosis-pathogenesis-and-clinical-features-in-patients-undergoing-dialysis-trea%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222015-03-21T01%3A08%3A42Z%22%7D%7D%2C%7B%22key%22%3A%2275IASB8J%22%2C%22library%22%3A%7B%22id%22%3A2147300%7D%2C%22meta%22%3A%7B%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EPress%20Announcements%20-%20FDA%20authorizes%20use%20of%20first%20device%20to%20treat%20patients%20with%20dialysis-related%20amyloidosis.%20Accessed%20March%2018%2C%202015.%20%3Ca%20href%3D%27http%3A%5C%2F%5C%2Fwww.fda.gov%5C%2FNewsEvents%5C%2FNewsroom%5C%2FPressAnnouncements%5C%2Fucm436864.htm%27%3Ehttp%3A%5C%2F%5C%2Fwww.fda.gov%5C%2FNewsEvents%5C%2FNewsroom%5C%2FPressAnnouncements%5C%2Fucm436864.htm%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Press%20Announcements%20-%20FDA%20authorizes%20use%20of%20first%20device%20to%20treat%20patients%20with%20dialysis-related%20amyloidosis%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22The%20U.S.%20Food%20and%20Drug%20Administration%20today%20authorized%20use%20of%20Lixelle%20Beta%202-microglobulin%20Apheresis%20Column%2C%20the%20first%20device%20to%20treat%20dialysis-related%20amyloidosis%20%28DRA%29.%22%2C%22date%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fwww.fda.gov%5C%2FNewsEvents%5C%2FNewsroom%5C%2FPressAnnouncements%5C%2Fucm436864.htm%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222015-03-18T09%3A10%3A03Z%22%7D%7D%5D%7D